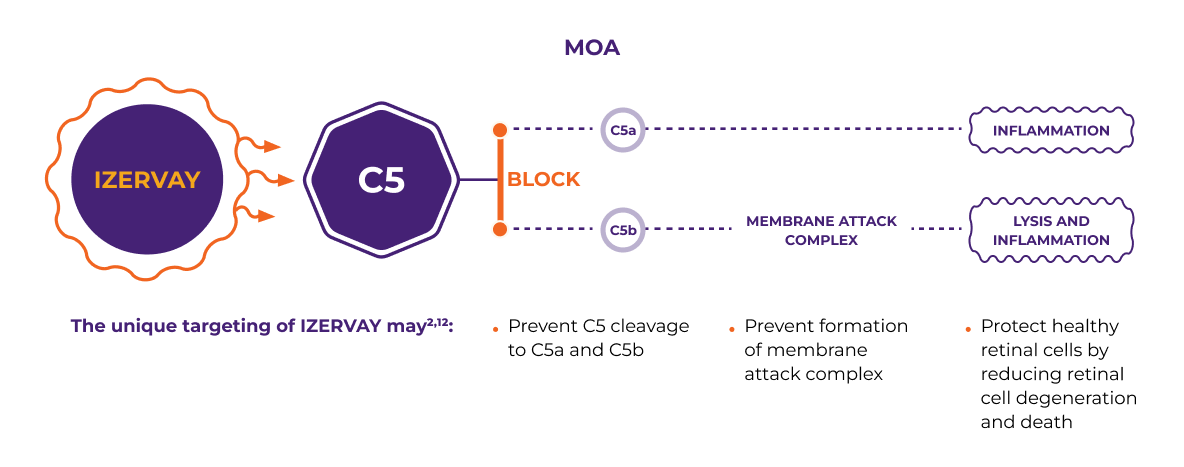
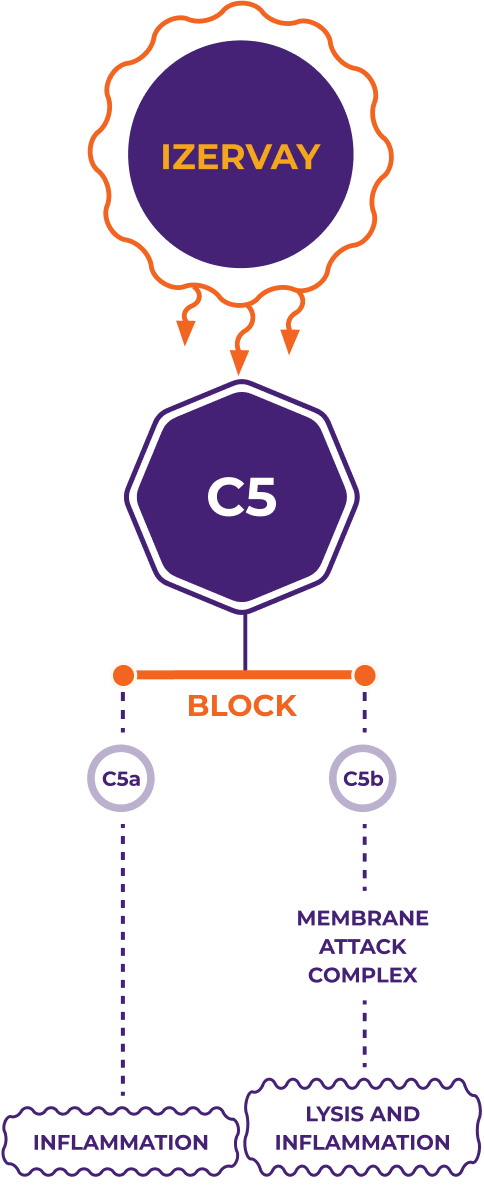
Only IZERVAY inhibits C5, which may reduce inflammation and protect healthy retina cells2,12
IZERVAY: Purposefully designed
MOA
The unique targeting of IZERVAY may2,12:
- Prevent C5 cleavage to C5a and C5b
- Prevent formation of membrane attack complex
- Protect healthy retinal cells by reducing retinal cell degeneration and death
RNA aptamer technology13
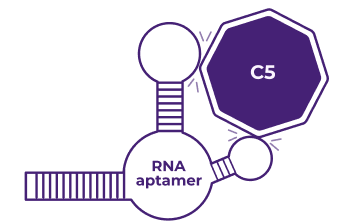
High binding affinity
Strong and specific inhibition of the target

Low immunogenicity
Low likelihood of inducing an immune response
Comprehensive packaging for seamless administration2

Preparation and administration
IZERVAY can be administered with a 30-gauge injection needle, which may decrease patient discomfort.2,14
For added convenience in your practice, unopened vials of IZERVAY can remain unrefrigerated for up to 24 hours.2
GA=geographic atrophy; RNA=ribonucleic acid.
Find out what your peers are saying about treating GA with IZERVAY
Next pageIMPORTANT SAFETY INFORMATION AND INDICATION
CONTRAINDICATIONS
IZERVAY® is contraindicated in patients with ocular or periocular infections and in patients with active intraocular inflammation.
WARNINGS AND PRECAUTIONS
Endophthalmitis and Retinal Detachments
- Intravitreal injections, including those with IZERVAY, may be associated with endophthalmitis and retinal detachments. Proper aseptic injection technique must always be used when administering IZERVAY in order to minimize the risk of endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay and should be managed appropriately.
Neovascular AMD
- In clinical trials, use of IZERVAY was associated with increased rates of neovascular (wet) AMD or choroidal neovascularization (7% when administered monthly and 4% in the sham group) by Month 12. Over 24 months, the rate of neovascular (wet) AMD or choroidal neovascularization in the GATHER2 trial was 12% in the IZERVAY group and 9% in the sham group. Patients receiving IZERVAY should be monitored for signs of neovascular AMD.
Increase in Intraocular Pressure
- Transient increases in intraocular pressure (IOP) may occur after any intravitreal injection, including with IZERVAY. Perfusion of the optic nerve head should be monitored following the injection and managed appropriately.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5%) reported in patients receiving IZERVAY were conjunctival hemorrhage, increased IOP, blurred vision, and neovascular age-related macular degeneration.
INDICATION
IZERVAY (avacincaptad pegol intravitreal solution) is indicated for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD)
Please see full Prescribing Information for more information.
To request medical information, please call 1-800-727-7003 or send an email to medinfo.americas@astellas.com. To report an adverse event or product complaint, please call 1-800-727-7003 or send an email to safety-us@astellas.com.
IMPORTANT SAFETY INFORMATION AND INDICATION
CONTRAINDICATIONS
IZERVAY® is contraindicated in patients with ocular or periocular infections and in patients with active intraocular inflammation.
WARNINGS AND PRECAUTIONS
Endophthalmitis and Retinal Detachments
- Intravitreal injections, including those with IZERVAY, may be associated with endophthalmitis and retinal detachments. Proper aseptic injection technique must always be used when administering IZERVAY in order to minimize the risk of endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay and should be managed appropriately.
Neovascular AMD
- In clinical trials, use of IZERVAY was associated with increased rates of neovascular (wet) AMD or choroidal neovascularization (7% when administered monthly and 4% in the sham group) by Month 12. Over 24 months, the rate of neovascular (wet) AMD or choroidal neovascularization in the GATHER2 trial was 12% in the IZERVAY group and 9% in the sham group. Patients receiving IZERVAY should be monitored for signs of neovascular AMD.
Increase in Intraocular Pressure
- Transient increases in intraocular pressure (IOP) may occur after any intravitreal injection, including with IZERVAY. Perfusion of the optic nerve head should be monitored following the injection and managed appropriately.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5%) reported in patients receiving IZERVAY were conjunctival hemorrhage, increased IOP, blurred vision, and neovascular age-related macular degeneration.
INDICATION
IZERVAY (avacincaptad pegol intravitreal solution) is indicated for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD)
Please see full Prescribing Information for more information.
To request medical information, please call 1-800-727-7003 or send an email to medinfo.americas@astellas.com. To report an adverse event or product complaint, please call 1-800-727-7003 or send an email to safety-us@astellas.com.