The majority of patients with GA want treatment to preserve their vision for longer3

In a claims analysis of newly diagnosed patients with GA,
~14%
of patients are being treated5*

Yet in a survey of people with self-reported GA,
84%
said they would try a treatment to slow their GA and preserve their vision for longer3†
GA requires a different mindset
It's about taking action now, so you can protect healthy retinal cells which may preserve vision as long as possible1,2
My goal as a physician is to preserve that vision for as long as I can, and I think it’s crucial to intervene early.”
– Dr. Arshad M. Khanani, Vitreoretinal Surgeon
I want to maintain my eyesight for as long as possible, and I felt a real sense of urgency to find a treatment that may help that.”
– Ken, real GA patient


Step into the role of protector today
When evaluating GA patients for treatment
Prioritize those at risk for accelerated progression1
The sooner you act, the more you may preserve for longer.1,2 See below for risk factors associated with faster progression of GA:
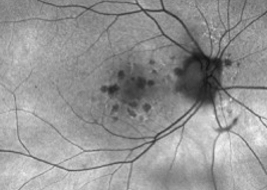
Multifocal lesions often grow faster than unifocal lesions6
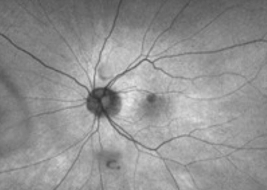
Extrafoveal lesions often progress faster than foveal lesions6
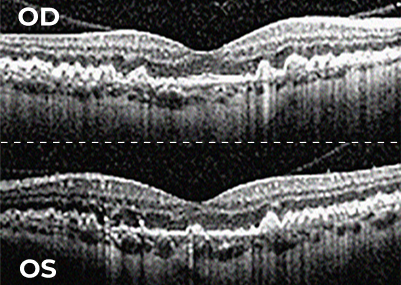
Patients with bilateral GA often progress faster than those with unilateral GA6,7
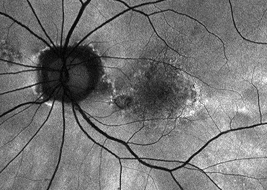
Hyperfluorescence on FAF imaging is often correlated with faster lesion progression6

Proactively protecting healthy retinal cells for longer is essential, especially in those at risk of faster progression. Patients appreciate this early management.1,2 It resonates with them if you are doing everything you can.”
– Dr. David S. Chin Yee, Retina Specialist
Quotes reflect patient experience at the time of printing. Physicians featured have been compensated.
FAF=fundus autofluorescence; GA=geographic atrophy; OD=oculus dexter; OS=oculus sinister.
*Data from Symphony Health, AMD Claims (142,301 newly diagnosed GA patients) June 1, 2020, to July 31, 2025.5
†Results from a telephone survey of 203 individuals with self-reported GA. Participants were compensated for their time, and the survey was sponsored by a pharmaceutical company.3
See efficacy & safety data
Next pageIMPORTANT SAFETY INFORMATION AND INDICATION
CONTRAINDICATIONS
IZERVAY® is contraindicated in patients with ocular or periocular infections and in patients with active intraocular inflammation.
WARNINGS AND PRECAUTIONS
Endophthalmitis and Retinal Detachments
- Intravitreal injections, including those with IZERVAY, may be associated with endophthalmitis and retinal detachments. Proper aseptic injection technique must always be used when administering IZERVAY in order to minimize the risk of endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay and should be managed appropriately.
Neovascular AMD
- In clinical trials, use of IZERVAY was associated with increased rates of neovascular (wet) AMD or choroidal neovascularization (7% when administered monthly and 4% in the sham group) by Month 12. Over 24 months, the rate of neovascular (wet) AMD or choroidal neovascularization in the GATHER2 trial was 12% in the IZERVAY group and 9% in the sham group. Patients receiving IZERVAY should be monitored for signs of neovascular AMD.
Increase in Intraocular Pressure
- Transient increases in intraocular pressure (IOP) may occur after any intravitreal injection, including with IZERVAY. Perfusion of the optic nerve head should be monitored following the injection and managed appropriately.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5%) reported in patients receiving IZERVAY were conjunctival hemorrhage, increased IOP, blurred vision, and neovascular age-related macular degeneration.
INDICATION
IZERVAY (avacincaptad pegol intravitreal solution) is indicated for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD)
Please see full Prescribing Information for more information.
To request medical information, please call 1-800-727-7003 or send an email to medinfo.americas@astellas.com. To report an adverse event or product complaint, please call 1-800-727-7003 or send an email to safety-us@astellas.com.
IMPORTANT SAFETY INFORMATION AND INDICATION
CONTRAINDICATIONS
IZERVAY® is contraindicated in patients with ocular or periocular infections and in patients with active intraocular inflammation.
WARNINGS AND PRECAUTIONS
Endophthalmitis and Retinal Detachments
- Intravitreal injections, including those with IZERVAY, may be associated with endophthalmitis and retinal detachments. Proper aseptic injection technique must always be used when administering IZERVAY in order to minimize the risk of endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay and should be managed appropriately.
Neovascular AMD
- In clinical trials, use of IZERVAY was associated with increased rates of neovascular (wet) AMD or choroidal neovascularization (7% when administered monthly and 4% in the sham group) by Month 12. Over 24 months, the rate of neovascular (wet) AMD or choroidal neovascularization in the GATHER2 trial was 12% in the IZERVAY group and 9% in the sham group. Patients receiving IZERVAY should be monitored for signs of neovascular AMD.
Increase in Intraocular Pressure
- Transient increases in intraocular pressure (IOP) may occur after any intravitreal injection, including with IZERVAY. Perfusion of the optic nerve head should be monitored following the injection and managed appropriately.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5%) reported in patients receiving IZERVAY were conjunctival hemorrhage, increased IOP, blurred vision, and neovascular age-related macular degeneration.
INDICATION
IZERVAY (avacincaptad pegol intravitreal solution) is indicated for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD)
Please see full Prescribing Information for more information.
To request medical information, please call 1-800-727-7003 or send an email to medinfo.americas@astellas.com. To report an adverse event or product complaint, please call 1-800-727-7003 or send an email to safety-us@astellas.com.
